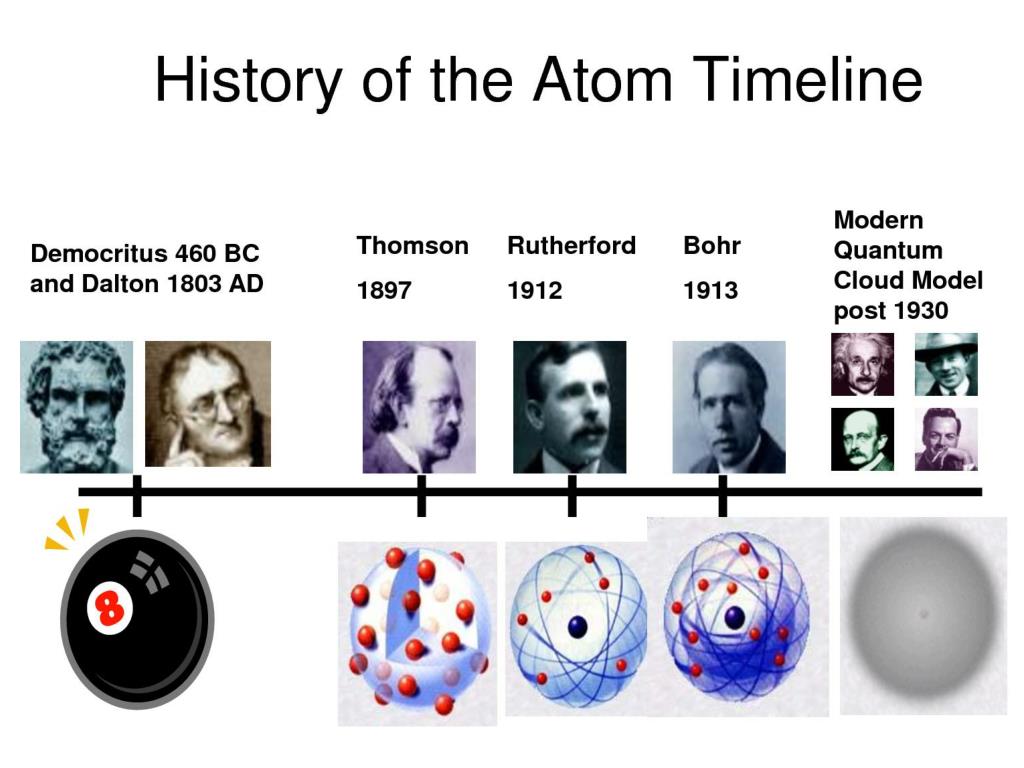
This weight never varied, so that any one atom of iron is equal to any other atom of iron. His chemical experiments led him to the conclusion that all matter in the universe was made up of a number of elements, such as hydrogen, iron, mercury and oxygen, and that each element consisted of indivisible atoms, and that the atom of each elementary substance had its own particular weight. It was in this sense that the word was used by the ancient Greeks for their atomic theories of the universe. The word “atom” originally meant a fundamental particle, or something which cannot be broken up into smaller parts. It was his interest in weather which led to his study of the air, and then of gases, and so to the formation of his Atomic Theory, for which he is most remembered. He began a meteorological diary in 1787 which he entered up meticulously every day for 57 years, making in all over 200,000 separate observations. 
His other great interest, which stayed with him all his life, was the science of weather. He, of course, was unaware of being in any way conspicuous. There is a story that Dalton unwittingly attracted attention to himself at a meeting of the British Association, to which he had been elected, by wearing the flaming red robes of a Doctor of Civil Law, when all about him were garbed in solemn black. He could recognise blue and shades of yellow, but little else. No doubt such defects in vision had existed since the beginning of time, but Dalton was the first man to remark upon it and write about its characteristics. He could not, at the time, understand why this was so but it was a problem that continued to occupy his mind.Īt the age of 28, he read a paper before the Manchester Literary and Philosophical Society, of which he was later to become secretary, on the nature of colour blindness, or “Daltonism” as it was known. This he could not understand since to him the colours looked much the same as the sombre shade of the grass on which they paraded. As a boy he was at a military review and heard onlookers comment on the brilliant colours of the soldiers’ uniforms. One of the first discoveries Dalton made was of his own colour blindness. This lasted until 1793 when he moved to Manchester, where he was to remain for the rest of his life, first to work in a college and then to become a freelance teacher of mathematics and science. Dalton worked as an assistant master, and when Bewley retired in 1785 became joint head of the school with his brother Jonathan, who had joined him. Finally, he left his native village in 1781 at the invitation of his cousin George Bewley, who kept a school at Kendal. He had a brief spell working on a farm, for which he was quite obviously unsuited. It had earned him an average of five shillings a week and there seemed no chance of improvement. His time as a teacher ended when he was 14. He liked always to think things out for himself, disdaining the advice and findings of others, claiming that he would have nothing to do with anything which he could not know and test from his own experience. Apart from some tuition from a distant relative and help from John Gough, a blind philosopher who encouraged his thirst for scientific knowledge, Dalton was self-taught. What little schooling he had ceased when he was 12, at which age he graduated from the position of pupil and took over as teacher at the little village school.ĭalton was possessed by an unusually studious nature and a patient application to matters of detail which marked him out as a natural mathematician. 
His family were Quakers and, throughout his life, Dalton remained true to his beliefs, taking his place at the Society of Friends’ meeting house twice a day on Sundays. He was born in 1766 in a small Cumberland village called Eaglesfield, where his father was a poor hand-loom weaver. John Dalton was curious about every subject under the sun. John Dalton, founder of chemical atomic theory by Peter Jackson
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
